RIVER BASINS
- A river basin is the portion of land drained by a river and its tributaries. It encompasses all of the land surface dissected and drained by many streams and creeks that flow downhill into one another, and eventually into one river. The final destination is an estuary or an ocean. As a bathtub catches all the water that falls within its sides, a river basin sends all the water falling on the surrounding land into a central river and out to the sea.
Everyone lives in a river basin. Even if we don't live near the water, we live on land that drains to a river or estuary or lake, and our actions on that land affect water quality and quantity far downstream. There are 17 river basins in North Carolina, draining 52,337 square miles of surface and underground waters. The topography of each basin determines the area that it drains, and whether that water - from creeks, rivers, springs, and aquifers - flows into the Atlantic Ocean or Gulf of Mexico.
When rain falls on your street, roof or yard, what creek or river will it flow through on its way downhill towards the sea? When you drain your bathtub or sink, where does the water go? When you turn on your tap to get a drink of water, where does the water come from?
As an artery connects the parts of a body to one another, so a river threads together the creeks and streams, valleys and hills, lakes and underground springs that share a common assembly of water. Whatever happens to surface or groundwater in one part of the river basin will find its way to other parts. If water is diverted out of its downward course in one section, other parts will come to "know" of its absence. A river basin comes closer than any other defined area of land, with the exception of an isolated island, to meeting the definition of an ecosystem in which all things, living and non-living, are connected and interdependent.

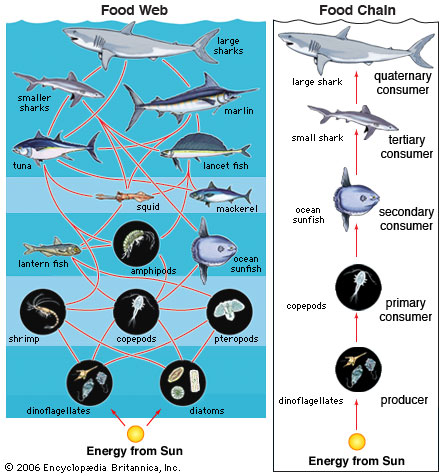
UPWELLING- the reason for upwelling in the ocean and fisheries is that Upwelling is an oceanographic phenomenon that involves wind-driven motion of dense, cooler, and usuallynutrient-rich water towards the ocean surface, replacing the warmer, usually nutrient-depleted surface water. The nutrient-rich upwelled water stimulates the growth and reproduction of primary producers such as phytoplankton.
ECOTOURISM- Tourism is the fastest growing division of the world economy and is responsible for more than 200 million jobs all over the world. In the US alone, tourism resulted in an economic gain of 478 billion dollars. With 700 million people traveling to another country in the year 2000, tourism is in the top five economic contributors to 83% of all countries and the most important economy for 38% of countries. The tourism industry is based on natural resources present in each country and usually negatively affect ecosystems because it is often left unmanaged. However, sustainable tourism can actually promote conservation of the environment.
Several tests can be performed on the water to ensure the water is fit for consumption. Some of these tests include Water Temperature, pH, Dissolved Oxygen (DO), Turbidity, Total Dissolved Solids (TDS) and Salinity, Water Hardness, andWater-Borne Microorganisms. Water Temperature: Water temperature can give us an idea of the overall water quality. Unusually high temperatures indicate that water quality has gone down. Warmer water has a smaller amount of dissolved oxygen (discussed later) and thus a higher chance that microorganisms are growing.
pH: pH is essentially a measure of acidity. pH values range between 0 and 14, with 7 being neutral. Below 7 indicates an acidic solution and above it is increasingly basic.
Chem Window --- pH
So why do we take pH readings of our drinking water? Since we must maintain our body pH at 7.4, highly basic or acidic water is not suitable for drinking.
How does water deviate from its pure pH of 7?
Even pure rain is naturally a little acidic. Water exposed to the atmosphere becomes slightly acidic because of dissolved CO2from the air.
H2O + CO2 <==> H2CO3 <==> HCO3- + H+In addition to the naturally occurring CO2 from the air, large amounts of organic waste matter in water will also increase the CO2 content, increasing acidity. The waste serves as food for microorganisms, which allows them to multiply and release CO2 as a product of respiration.
A great deal of the H+ ions come from acid rain, which is itself a product of polluted air. Industrial and automotive sources release large amounts of SO2, NOX, and CO2, which react in the atmosphere to produce acid rain.
Dissolved Oxygen (DO): Dissolved oxygen refers to the amount of oxygen gas (O2) that has dissolved into the water. This is an important test for water quality because oxygen levels reveal a great deal about the amount of pollutants in the water. In general, higher dissolved oxygen levels indicate higher water quality.
Abnormally low concentrations of oxygen, however, suggest that there is organic waste matter present in the water. Yuck! Tiny microorganisms feed on this waste matter and consume oxygen to stay alive. So more waste translates to more food for these microorganisms. More food means more microorganisms will grow and even more oxygen will be consumed.
ECOTOURISM- Tourism is the fastest growing division of the world economy and is responsible for more than 200 million jobs all over the world. In the US alone, tourism resulted in an economic gain of 478 billion dollars. With 700 million people traveling to another country in the year 2000, tourism is in the top five economic contributors to 83% of all countries and the most important economy for 38% of countries. The tourism industry is based on natural resources present in each country and usually negatively affect ecosystems because it is often left unmanaged. However, sustainable tourism can actually promote conservation of the environment.
Several tests can be performed on the water to ensure the water is fit for consumption. Some of these tests include Water Temperature, pH, Dissolved Oxygen (DO), Turbidity, Total Dissolved Solids (TDS) and Salinity, Water Hardness, andWater-Borne Microorganisms. Water Temperature: Water temperature can give us an idea of the overall water quality. Unusually high temperatures indicate that water quality has gone down. Warmer water has a smaller amount of dissolved oxygen (discussed later) and thus a higher chance that microorganisms are growing.
pH: pH is essentially a measure of acidity. pH values range between 0 and 14, with 7 being neutral. Below 7 indicates an acidic solution and above it is increasingly basic.
Chem Window --- pH
So why do we take pH readings of our drinking water? Since we must maintain our body pH at 7.4, highly basic or acidic water is not suitable for drinking.
How does water deviate from its pure pH of 7?
Even pure rain is naturally a little acidic. Water exposed to the atmosphere becomes slightly acidic because of dissolved CO2from the air.
H2O + CO2 <==> H2CO3 <==> HCO3- + H+In addition to the naturally occurring CO2 from the air, large amounts of organic waste matter in water will also increase the CO2 content, increasing acidity. The waste serves as food for microorganisms, which allows them to multiply and release CO2 as a product of respiration.
A great deal of the H+ ions come from acid rain, which is itself a product of polluted air. Industrial and automotive sources release large amounts of SO2, NOX, and CO2, which react in the atmosphere to produce acid rain.
Dissolved Oxygen (DO): Dissolved oxygen refers to the amount of oxygen gas (O2) that has dissolved into the water. This is an important test for water quality because oxygen levels reveal a great deal about the amount of pollutants in the water. In general, higher dissolved oxygen levels indicate higher water quality.
Abnormally low concentrations of oxygen, however, suggest that there is organic waste matter present in the water. Yuck! Tiny microorganisms feed on this waste matter and consume oxygen to stay alive. So more waste translates to more food for these microorganisms. More food means more microorganisms will grow and even more oxygen will be consumed.

HUMAN AFFECTS- humans have the oppourtunity to make sea life either better or worse, with ecotourism going on people are producing enough revenue iin order to keep the estuaries clean and safe, and the other side where people are constantly polluting and putting the ocean life in danger.
WATER TREATMENT- The amount and type of treatment applied by a PWS
varies with the source type and quality. Many ground
water systems can satisfy all federal requirements
without applying any treatment, while others need
to add chlorine or additional treatment. US EPA
is developing a ground water rule that will specify
the appropriate use of disinfection and will address
other components of ground water systems to assure
public health protection. Because surface water
systems are exposed to direct wet weather runoff
and to the atmosphere and are therefore more easily
contaminated, federal and state regulations require
that these systems treat their water. Disinfection
of drinking water is one of the major public
health advances of the 20th century. However, the
disinfectants themselves can react with naturally
occurring materials in the water to form unintended
byproducts which may pose health risks. A major
challenge for water suppliers is balancing the risks
from microbial pathogens and disinfection byproducts.
The Stage 1 Disinfectants and Disinfection Byproducts
Rule and the Interim Enhanced Surface Water
Treatment Rule together address these risks.
Water suppliers use a variety of treatment processes
to remove contaminants from drinking water. These
individual processes may be arranged in a “treatment
train” (a series of processes applied in sequence).
WATER TREATMENT- The amount and type of treatment applied by a PWS
varies with the source type and quality. Many ground
water systems can satisfy all federal requirements
without applying any treatment, while others need
to add chlorine or additional treatment. US EPA
is developing a ground water rule that will specify
the appropriate use of disinfection and will address
other components of ground water systems to assure
public health protection. Because surface water
systems are exposed to direct wet weather runoff
and to the atmosphere and are therefore more easily
contaminated, federal and state regulations require
that these systems treat their water. Disinfection
of drinking water is one of the major public
health advances of the 20th century. However, the
disinfectants themselves can react with naturally
occurring materials in the water to form unintended
byproducts which may pose health risks. A major
challenge for water suppliers is balancing the risks
from microbial pathogens and disinfection byproducts.
The Stage 1 Disinfectants and Disinfection Byproducts
Rule and the Interim Enhanced Surface Water
Treatment Rule together address these risks.
Water suppliers use a variety of treatment processes
to remove contaminants from drinking water. These
individual processes may be arranged in a “treatment
train” (a series of processes applied in sequence).